Whitepaper: How logistics are critical in clinical studies
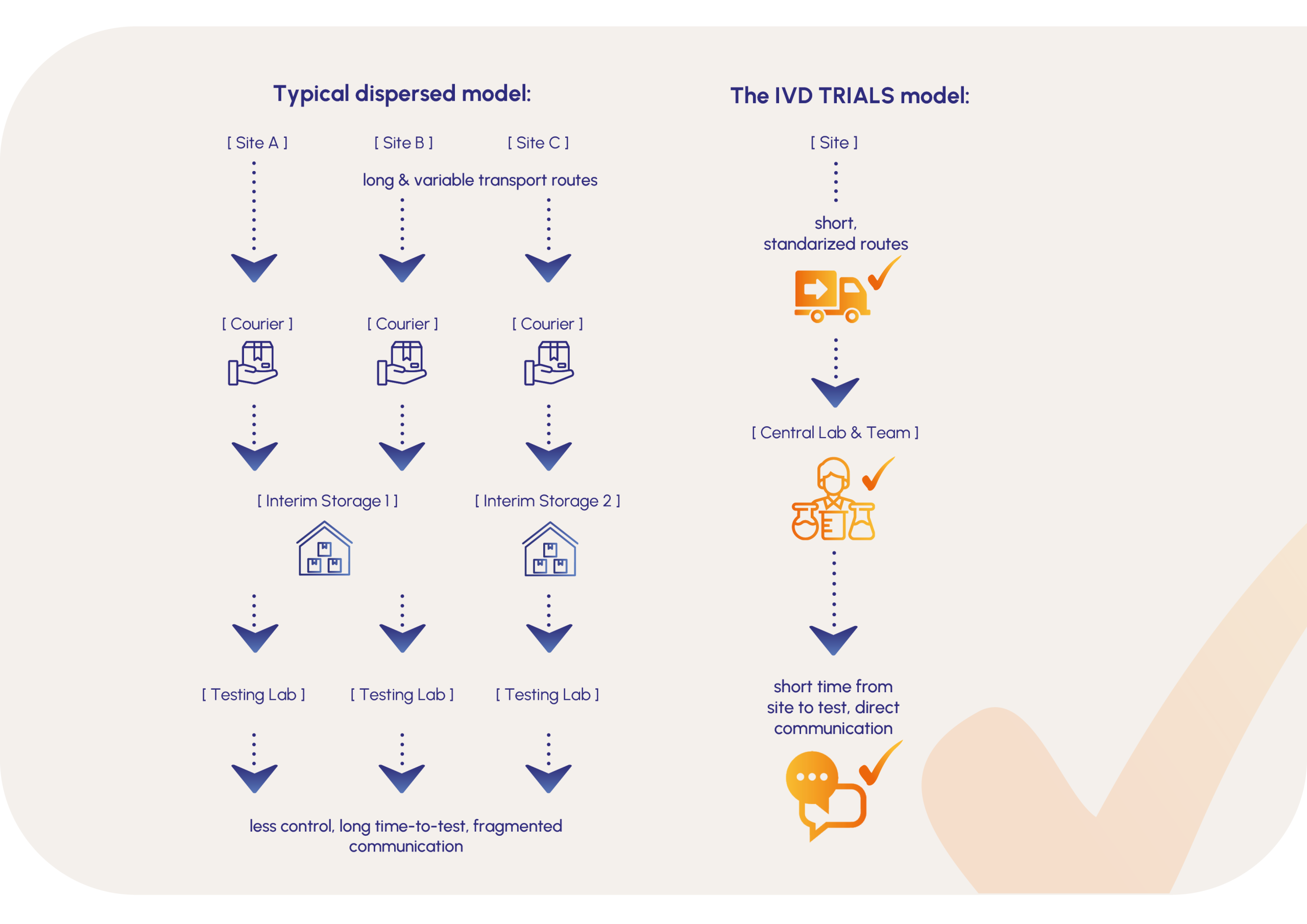
Many diagnostic developers underestimate how much logistics and operational setup influences IVD study timeline. A poorly coordinated study setup can extend timelines significantly and delay your time-to-market. That’s why choosing the right CRO partner matters.
In practice, delays are often not caused by scientific challenges, but by operational factors such as:
- Clinical trial logistics and supply coordination
- Sample handling and transport
- Communication between study partners and laboratories
Understanding how these operational elements impact timelines is essential for keeping IVD studies on schedule and avoiding costly delays.
In this whitepaper, we break down the key operational factors that influence study timelines, based on our experience supporting 300+ clinical studies.